Gold is not typically known for its flexibility, but at the nanoscale, a team of researchers has found that gold nanoparticles can behave more like a liquid than a solid, reorganizing themselves into entirely different structures in response to small changes in temperature or physical pressure.
A research team led by Dr. Rina Sato, formerly of Tohoku University and currently at the National Institute for Materials Science, and Professor Kiyoshi Kanie of Tohoku University, discovered that gold nanoparticles positioned at the boundary between air and water can dramatically shift their arrangement when conditions change. The study, reported by Phys.org, reveals for the first time that minor changes in how organic molecules are distributed on nanoparticle surfaces can drive large-scale structural transformations across an entire layer of nanoparticles.
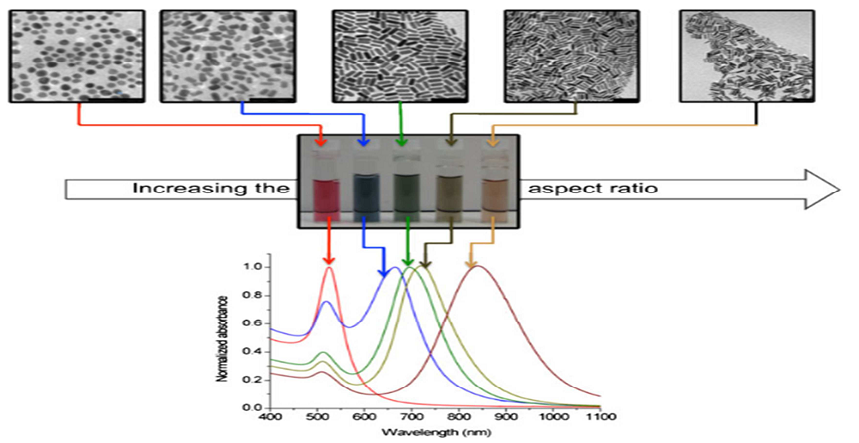
The behavior of nanoparticles matters because the optical, electronic, and magnetic properties of inorganic nanoparticles depend strongly on how they are arranged relative to one another. Being able to control that arrangement, and change it on demand, could give scientists a powerful tool for designing materials with tunable properties.
To study this, the team engineered gold nanoparticles coated with two types of organic molecules: a temperature-responsive dendritic liquid-crystal molecule called a dendron, and a simpler linear-chain ligand. They then placed these coated nanoparticles at the air and water interface, where nanoparticles with hydrophobic surface molecules naturally assemble into flat, two-dimensional layers.
What they observed was highly dynamic. At room temperature, the nanoparticles clustered into isolated island-like structures. As temperature climbed, those islands gradually reorganized into chain-like arrangements. At around 40 degrees Celsius, the chains gave way to large, network-like patterns spread across the layer. When the researchers mechanically compressed that network, it reversed course and returned to the island-like formations.
To understand the mechanism behind this reversible behavior, the team used X-ray measurements at the DESY synchrotron facility in Hamburg, Germany. The data showed that the two types of surface molecules were spontaneously redistributing themselves across each nanoparticle's surface in response to the external stimulus, changing the effective shape and symmetry of the particles and driving the collective reorganization of the entire assembly.
"This work demonstrates how very small molecular-level changes can lead to dramatic structural transformations in nanoparticle systems," said Kanie. "We believe this finding opens a new pathway for designing smart and adaptive materials that respond dynamically to their environment."
The research points toward a new strategy for building materials that can sense and respond to their surroundings, switching properties when needed without requiring complex external machinery. The ability to trigger these changes at temperatures as low as 40 degrees Celsius makes the approach particularly practical for real-world applications.